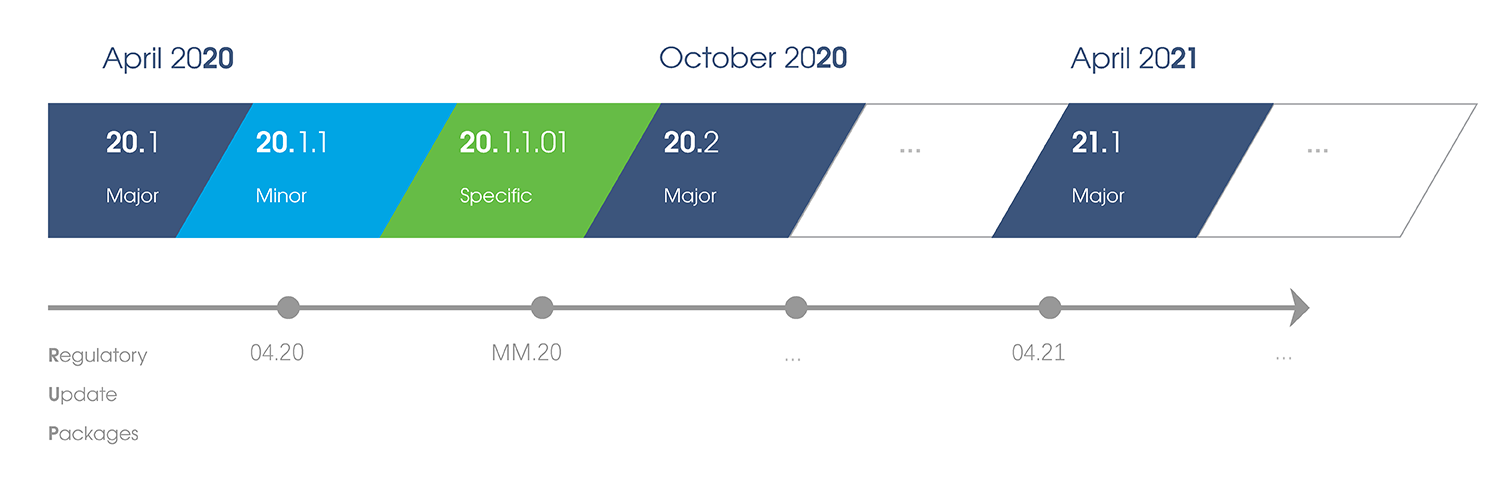
Since April 2018, LORENZ Life Sciences Group has started switching all of its products to a unified release naming system based on the calendar year. The company's continuous cycle of two major software releases each year - one in April and the other in October, each followed by minor releases and specific releases if necessary - remains unchanged.
LORENZ encourages all customers to update their software regularly. We typically support our major releases for at least two years, providing minor and specific releases for each throughout the entire period.
In addition, LORENZ offers Regulatory Update Packages. These packages address recent specification updates by regulatory authorities.
To stay informed about both major and minor releases as well as Regulatory Update Packages, please subscribe to Product & Service Update Announcements in your LORENZ ID.
Major Release
Two major releases take place each year on a regular schedule. These include substantial changes to the software or completely new components, and are communicated via LORENZ ID.
The first takes place every April and the second happens in October. These major releases of LORENZ products are all consistently labelled. The first two digits (xx) indicate the year, followed by .1 for the April release (xx.1) and .2 for the October one (xx.2).
Minor Release
A minor release typically includes changes such as support for a new specification or validation criteria. These are also communicated via LORENZ ID.
For minor releases, a third place is added to the version number. So, release xx.1.1 indicates the first minor release based on the April major release in year xx.
Specific Release
A specific release might include the delivery of language packs, or a fix for a specific issue. Typically, these are not communicated by LORENZ ID.
Specific releases each add a fourth (two-digit) number to the version designation. So, the first specific release based on the April major release in year xx would be xx.1.1.01.
Regulatory Update Packages
These packages address recent specification updates by regulatory authorities and allow to adapt the submission configuration without going through a software update process and full revalidation of the software. Regulatory Update Packages are communicated via LORENZ ID. More information on these packages can be found in the Administrator Guide.
